Research Interests
Thiol-redox Signaling and Neuronal Cell Death
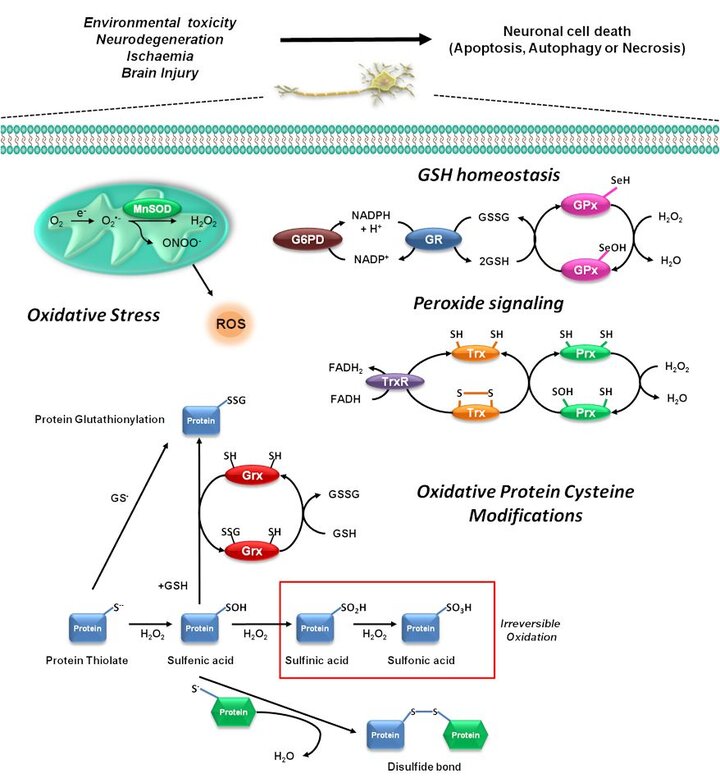
Alterations in the cellular redox balance regulate the activation of distinct signaling cascades leading to the progression of a variety of cell death programs. However, the exact mechanisms involved in the regulation of cell death by redox signaling are still far from being understood. In our research group we aim to elucidate the molecular mechanisms involved in the regulation of cell death progression by redox signaling. Because the intracellular thiol-disulfide (GSH/GSSG) balance is considered the major determinant of the redox status of the cell, we are particularly interested in studying how alterations in thiol homeostasis regulate the activation/inactivation of the cell death machinery during neuronal cell death.
To understand the exact regulatory role of oxidative stress in the signal transduction events leading to cell death we must determine the substrate specificity and biomolecular alterations caused by the reaction of reactive species. Proteins are likely to be the major targets for oxidative modification, as a result of their abundance in cells. Relative to other amino acids, cysteine residues are among the most sensitive to oxidation and an important cellular target or sensor of reactive species is the thiol group of the amino acid cysteine. Under mild oxidative stress, reversible oxidation of selective cysteine protein residues may serve as redox sensors and signal transducers for conveying cellular anti-stress responses. However, when encountering severe oxidative insults, cysteine thiols undergo irreversible oxidative modifications, causing protein degradation and cell death. We are interested in understanding the role of oxidative protein cysteine modifications (glutathionylateion, sulfenylation) in the regulation of the cell death machinery.
Cells defend themselves against ROS damage through specific enzymatic or non-enzymatic mechanisms. We are also interested in the role of thiol-oxidoreductases (peroxiredoxins, glutaredoxins and thioredoxins) regulating oxidative stress and cysteine oxidation during neuronal cell death.
Environmental Toxicity, Oxidative Stress and Cell Death Signaling
The environment represents a key contributor to human health and disease. Exposure to many toxicants such as metals and pesticides have detrimental effects on health and are considered to contribute substantially to a number of diseases of major public health significance. It has been recognized that many of the toxic effects induced by environmental stressors are mediated by regulation/induction of cell death and oxidative stress whose deregulation has been associated to several environmental diseases. The overall impact of environmental changes on the mechanisms of cell death progression is poorly understood yet the consequences of modifying/regulating them can result in a potential increased risk of developing diseases such as cancer and neurodegeneration, which are associated to alterations in cell death rates. We are interested in identifying the molecular mechanisms by which oxidative stress regulates cell death during environmental toxicity.
Brain Injury (Ischemia, Excitotoxicity and Edema)
Neuronal cell death is an important component of distinct pathological states including hyponatraemia, traumatic brain injury, ischemia and excytotoxic insults. Our research aims to understand the role of redox signaling in neuronal injury induced by hypoxic/ischemic conditions, excytotoxic insults and cellular stretch.
Cytotoxic edema is the result of a deregulated cell swelling of neurons and glia and the subsequent reduction in the extracellular space. We are particularly interested in characterizing the signaling events that modulate the activation of ionic/osmolyte efflux pathways under these circumstances.